Retatrutide-RUO 10mg
Price range: $90.00 through $600.00
Buy Retatrutide RUO 10mg from Profound Peptides — a Retatrutide research grade peptide independently verified at 99.914% purity by Freedom Diagnostics, a US-based third-party laboratory (COA: 2603130114, Lot: RET10-2162026).
Supplied as white lyophilized powder with a batch-specific, publicly verifiable Certificate of Analysis — one of the highest documented purity results available from any Retatrutide RUO 10mg supplier in the USA.
For in vitro laboratory and scientific research use only. Not for human consumption.
Retatrutide RUO 10mg — Full Product Specifications:
| Specification | Detail |
| Also Known As | LY3437943 · GLP RT · GGG Triple Agonist |
| CAS Number | 2381089-83-2 |
| Molecular Formula | C₂₂₁H₃₄₂N₄₆O₆₈ |
| Molecular Weight | ~4,731 Da |
| Sequence Length | 39 amino acids |
| Form | White lyophilized powder |
| Purity — this batch | 99.914% — Freedom Diagnostics verified |
| Testing Method | HPLC + UV Detection + Mass Spectrometry (LCMS/MS) |
| Lot Number | RET10-2162026 |
| COA Accession | 2603130114 — publicly verifiable at FreedomDiagnosticsTesting.com |
| Receptor Targets | GLP-1R · GIPR · GCGR (triple agonist) |
| Half-life | ~144 hours (~6 days) in relevant research models |
| Long-term storage | -20°C · sealed vial · up to 24 months |
| Short-term storage | 4°C · up to 4–6 weeks reconstituted |
| Reconstitution | Bacteriostatic water (BAC water) recommended |
| Intended Use | In vitro laboratory research only — not for human use |
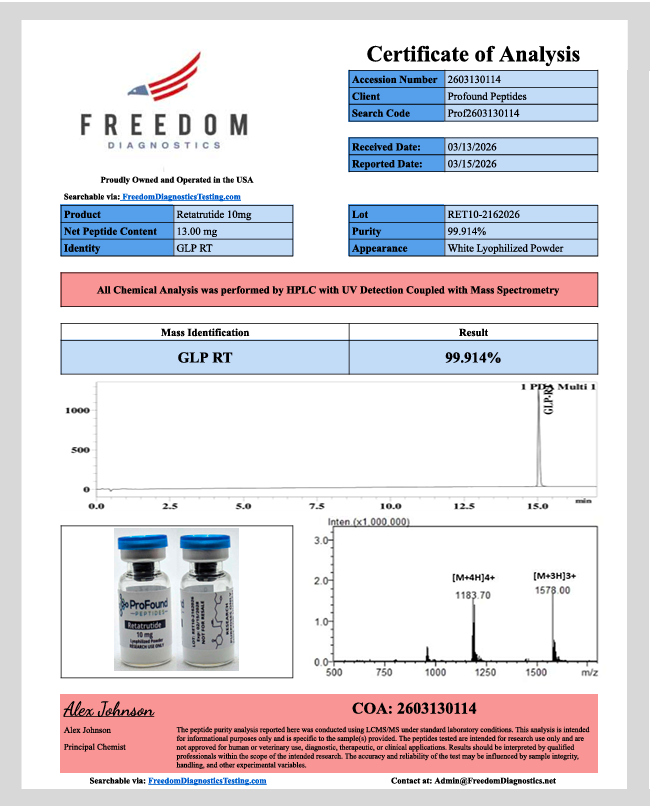
Certificate of Analysis — Freedom Diagnostics
Every batch is independently verified by a US-owned laboratory. Publicly searchable — no involvement from Profound Peptides required.
| PURITY RESULT | 99.914% |
| TESTING LAB | Freedom Diagnostics (USA) |
| LOT NUMBER | RET10-2162026 |
| COA/ACCESSION | 2603130114 |
| TESTING METHOD | HPLC + Mass Spec (LCMS/MS) |
| NET PEPTIDE CONTENT | 13.00mg confirmed |
| REPORT DATE | March 15, 2026 |
| PRINCIPAL CHEMIST | Alex Johnson |
How Retatrutide Works — Triple Receptor Mechanism
Retatrutide (LY3437943) is a high purity peptide that simultaneously activates three hormone receptors: GLP-1R, GIPR, and GCGR. This triple agonism — referred to in research literature as a GGG triagonist — is what separates it from Semaglutide (GLP-1R only) and Tirzepatide (GLP-1R + GIPR).
GLP-1 Receptor (GLP-1R)
Drives insulin secretion in a glucose-dependent manner, suppresses glucagon release, slows gastric emptying, and signals satiety through central nervous system pathways.
GIP Receptor (GIPR)
Augments insulin secretion and plays a role in lipid storage and metabolism. Combined GLP-1R and GIPR activation produces synergistic glycemic and metabolic effects beyond either receptor alone.
Glucagon Receptor (GCGR)
The unique differentiator. GCGR activation stimulates thermogenesis through brown adipose tissue, promotes hepatic fat oxidation, increases basal metabolic rate, and contributes to lipolysis — creating energy expenditure effects unavailable through GLP-1R and GIPR activation alone.
The structural basis for this activity is a 39-amino-acid GIP-backbone sequence with non-natural amino acid substitutions (Aib at positions 2 and 20) for helix stabilization, and a C20 fatty diacid acylation at Lys¹⁷ that enables reversible albumin binding — giving Retatrutide lyophilized powder a half-life of ~144 hours (~6 days) in relevant research models.
Research Applications
The following are primary in vitro and preclinical research applications for scientific research Retatrutide. All are for laboratory use only.
- Metabolic disorder research: Obesity models, insulin resistance, type 2 diabetes — coordinated multi-pathway signaling across GLP-1R, GIPR, and GCGR simultaneously.
- Energy expenditure & thermogenesis: GCGR-driven brown adipose tissue activation and basal metabolic rate modulation — the defining research application for Retatrutide for metabolic research.
- Hepatic steatosis (MASH/NAFLD): Glucagon receptor agonism promotes hepatic fat oxidation — relevant to liver fat accumulation research.
- Glucose homeostasis: Dual incretin component (GLP-1R + GIPR) for studying glucose-dependent insulin secretion mechanisms.
- Receptor crosstalk studies: Investigating synergistic (vs additive) effects of tri-receptor co-activation — a frontier area in incretin biology.
- Comparative agonist research: Well-characterized receptor binding profile (GLP-1R EC₅₀ ~0.775 nM, GIPR EC₅₀ ~0.0643 nM, GCGR EC₅₀ ~5.79 nM) for controlled comparative assay design.
Retatrutide vs Tirzepatide vs Semaglutide — Research Comparison
For researchers selecting compounds for metabolic signaling studies. All three are available as RUO research peptides.
| Property | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Agonist class | Single (GLP-1R) | Dual (GLP-1R + GIPR) | Triple (GLP-1R + GIPR + GCGR) |
| GCGR activation | No | No | Yes — thermogenesis + fat oxidation |
| CAS number | 910463-68-2 | 2023788-19-2 | 2381089-83-2 |
| Phase 2 weight loss | ~14.9% at 68 wks | ~20–22% at 72 wks | ~24.2% at 48 wks / 28.7% at 68 wks |
| Research status (2026) | FDA approved (Rx) | FDA approved (Rx) | Phase 3 TRIUMPH trials (2026) |
Storage & Reconstitution Guide
Storage
- Long-term: -20°C, sealed vial, up to 24 months. Away from light and moisture.
- Short-term: 4°C once reconstituted. Use within 4–6 weeks.
- Avoid freeze-thaw cycles: Aliquot into single-use volumes before long-term storage.
Reconstitution
- Solvent: Bacteriostatic water (BAC water) recommended. Sterile saline (0.9% NaCl) is acceptable.
- Method: Add solvent slowly along the inside vial wall. Swirl gently — do not vortex or shake.
- Volume: Vial accommodates up to 3mL. Adjust to your required working concentration.
- Do not refreeze: Once reconstituted, store at 4°C and do not refreeze.
Note: Peptides are supplied by mass (mg), not volume. The lyophilized powder is a thin layer or cake at the bottom of the vial — this is normal.
Published Research — Scientific References
Key published studies on Retatrutide (LY3437943) for research context. Provided for informational purposes only. This product is for laboratory research use only.
- NEJM Phase 2 (2023): Jastreboff AM et al. — up to 24.2% mean body weight reduction at 48 weeks. N Engl J Med 389(6):514–526. PMID: 37490471
- Lancet Phase 1b (2022): Urva S et al. — Safety and tolerability in type 2 diabetes. Lancet 400(10366):1869–1881. PMID: 36436526
- Cell Metabolism — Discovery (2022): Coskun T et al. — Mechanism and clinical proof of concept. Cell Metab 34(9):1234–1247. PMID: 36070788
- Phase 3 TRIUMPH Trials (ongoing): NCT05929066 and related registrations. Completion expected 2026–2027.
- Preclinical comparative (2025): Ma T et al. — Superior metabolic outcomes vs Tirzepatide and Liraglutide in db/db mice. Endocrinology 166(2):89–102.
Retatrutide RUO 10mg research peptide is intended exclusively for in vitro research and scientific experimentation. It is utilized in preclinical laboratory models to investigate multi-receptor agonism mechanisms, synergistic effects on metabolic signaling pathways, energy expenditure modulation (via GCGR), appetite regulation and satiety signaling (via GLP-1R and GIPR), glucose homeostasis, lipid metabolism, body weight dynamics, incretin/glucagon crosstalk, and related processes in cellular assays, tissue models, or animal systems exploring metabolic disorders, obesity models, and neuroendocrine regulation.





Reviews
There are no reviews yet.